Ligand-induced cell adhesion was discovered at our laboratory as one of the mechanisms underlying the synaptogenic effects of GDNF and its receptor GFRa1 (Ledda et al. Nat. Neurosci. 2007). GDNF treatment of cells expressing GFRa1 induces the formation of cell aggregates in a saturable process that requires continuous presence of GFRa1 on the cell membrane. Due to the presence of GFRa1 in pre- and post-synaptic specializations, and its requirement for synapse formation in vitro and in vivo, this novel cell adhesion activity may contribute to synapse formation and neuronal connectivity. Although the precise molecular mechanism (whether trans-homodimerization, receptor allosterism, or other) is still unclear, it represented the first example of cell adhesion mediated by binding of a ligand to a cell surface receptor which otherwise lacks cell adhesion activity. Now a group of researchers led by Prof. Masayoshi Mishina from the Uiversity of Tokyo have discovered a second example of this phenomenon, suggesting that it may be a general mechanism of cell-cell engagement. The new work involves the synaptogenic actions of GluRd2 and Neurexin receptors in cooperation with the Cerebellin 1 (Cbln1) ligand and has been published in the June 11 issue of Cell.
|
|||||
|
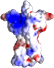
After quite some perseverance and few years of work, the 3D structure of the N-terminal portion of the extracellular domain (ECD) of the RET receptor has been determined by former postdoc fellow Svend Kjaer working at the lab of Nel McDonald in CRUK, London. The paper has appeared online in Nature Structure and Molecular Biology. During his tenure at our lab in 1998-2004, Svend studied RET intensively and published several papers on its biosynthesis, oligomerization and structure-function relationships. He spent several years producing fragments of the RET extracellular domain for structural characterization. Although sufficient for biochemical analyses, the yield was never enough for crystallization. The breakthrough at CRUK came after introduction of a few mutations, including two that knocked-off a couple of unpaired cysteines. The new structure gives tantalizing insights into how RET interacts with the GDNF ligand and co-receptor GFRa1 and how its mutation causes disease. |
|||||
|
Copyright © 2025 Carlos Ibanez Lab @ KI - All Rights Reserved Powered by WordPress & Atahualpa |
|||||