|
|
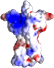
February 21st, 2024 Genome-wide studies have identified three missense variants in the human gene ACVR1C, encoding the TGF-β superfamily receptor ALK7, that correlate with altered waist-to-hip ratio adjusted for body mass index (WHR/BMI), a measure of body fat distribution.
In our latest paper, to move from correlation to causation and understand the effects of these variants on fat accumulation and adipose tissue function, we introduced each of the variants in the mouse Acvr1c locus and investigated metabolic phenotypes in comparison with a null mutation.
Mice carrying the I195T variant showed resistance to high fat diet (HFD)-induced obesity, increased catecholamine-induced adipose tissue lipolysis and impaired ALK7 signaling, phenocopying the null mutants. Mice with the I482V variant displayed an intermediate phenotype, with partial resistance to HFD-induced obesity, reduction in subcutaneous, but not visceral, fat mass, decreased systemic lipolysis and reduced ALK7 signaling. Surprisingly, mice carrying the N150H variant were metabolically indistinguishable from wild type under HFD, although ALK7 signaling was reduced at low ligand concentrations.
Together, these results validate ALK7 as an attractive drug target in human obesity and suggest a lower threshold for ALK7 function in humans compared to mice.
The paper has been published in Molecular Metabolism
January 22nd, 2024 How receptors juggle their interactions with multiple downstream effectors remains poorly understood.
In our latest paper, we report that the outcome of death receptor p75NTR signaling is determined through competition of effectors for interaction with its intracellular domain, in turn dictated by the nature of the ligand. While NGF induces release of RhoGDI through recruitment of RIP2, thus decreasing RhoA activity in favor of NFkB signaling, MAG induces PKC-mediated phosphorylation of the RhoGDI N-terminus, promoting its interaction with the juxtamembrane domain of p75NTR, disengaging RIP2, and enhancing RhoA activity in detriment of NF-kB. This results in stunted neurite outgrowth and apoptosis in cerebellar granule neurons. If presented simultaneously, MAG prevails over NGF. The NMR solution structure of the complex between the RhoGDI N-terminus and p75NTR juxtamembrane domain reveals previously unknown structures of these proteins and clarifies the mechanism of p75NTR activation.
These results show how ligand-directed competition between RIP2 and RhoGDI for p75NTR engagement determine axon growth and neuron survival. Similar principles are likely at work in other receptors engaging multiple effectors and signaling pathways.
The paper has been published in EMBO Reports
December 21st, 2022 Adipocyte hyperplasia and hypertrophy are the two main processes contributing to adipose tissue expansion, yet the mechanisms that regulate and balance their involvement in obesity are incompletely understood. Activin B/GDF-3 receptor ALK7 is expressed in mature adipocytes and promotes adipocyte hypertrophy upon nutrient overload by suppressing adrenergic signaling and lipolysis. In contrast, the role of ALK4, the canonical pan-activin receptor, in adipose tissue is unknown.
In our latest paper, we report that, unlike ALK7, ALK4 is preferentially expressed in adipocyte precursors, where it suppresses differentiation, allowing proliferation and adipose tissue expansion. ALK4 expression in adipose tissue increases upon nutrient overload and positively correlates with fat depot mass and body weight, suggesting a role in adipose tissue hyperplasia during obesity. Mechanistically, ALK4 signaling suppresses expression of CEBPα and PPARγ, two master regulators of adipocyte differentiation. Conversely, ALK4 deletion enhances CEBPα/PPARγ expression and induces premature adipocyte differentiation, which can be rescued by CEBPα knockdown.
These results clarify the function of ALK4 in adipose tissue and highlight the contrasting roles of the two activin receptors in the regulation of adipocyte hyperplasia and hypertrophy during obesity.
The paper has been published in The Journal Of Biological Chemistry
July 1st, 2022 ΔfosB is an alternatively spliced product of the FosB gene that is essential for dopamine-induced reward pathways and that acts as a master switch for addiction. However, the molecular mechanisms of its generation and regulation by dopamine signaling are unknown.
In this new paper, we report that dopamine D1 receptor signaling synergizes with the activin/ALK4/Smad3 pathway to potentiate the generation of ΔFosB mRNA in medium spiny neurons (MSNs) of the nucleus accumbens (NAc) via activation of the RNA-binding protein PCBP1, a regulator of mRNA splicing. Concurrent activation of PCBP1 and Smad3 by D1 and ALK4 signaling induced their interaction, nuclear translocation, and binding to sequences in exon-4 and intron-4 of FosB mRNA. Ablation of either ALK4 or PCBP1 in MSNs impaired ΔFosB mRNA induction and nuclear translocation of ΔFosB protein in response to repeated co-stimulation of D1 and ALK4 receptors. Finally, ALK4 is required in NAc MSNs of adult mice for behavioral sensitization to cocaine.
These findings uncover an unexpected mechanism for ΔFosB generation and drug-induced sensitization through convergent dopamine and ALK4 signaling.
The paper has been published in The EMBO Journal
November 8th, 2021 The medial habenula (mHb) is an understudied small brain nucleus linking forebrain and midbrain structures controlling anxiety and fear behaviors. The mechanisms that maintain the structural and functional integrity of mHb neurons and their synapses remain unknown.
In this study, we used spatiotemporally controlled Cre-mediated recombination in adult mice, and found that the glial cell–derived neurotrophic factor receptor alpha 1 (GFRα1) is required in adult mHb neurons for synaptic stability and function. mHb neurons express some of the highest levels of GFRα1 in the mouse brain, and acute ablation of GFRα1 results in loss of septo-habenular and habenulo-interpeduncular glutamatergic synapses, with the remaining synapses displaying reduced numbers of presynaptic vesicles. Chemo- and optogenetic studies in mice lacking GFRα1 revealed impaired circuit connectivity, reduced AMPA receptor postsynaptic currents, and abnormally low rectification index of AMPARs, suggesting reduced Ca2+ permeability. Further biochemical and proximity ligation assay studies defined the presence of GluA1/GluA2 (Ca2+ impermeable) as well as GluA1/GluA4 (Ca2+ permeable) AMPAR complexes in mHb neurons, as well as clear differences in the levels and association of AMPAR subunits with mHb neurons lacking GFRα1. Finally, acute loss of GFRα1 in adult mHb neurons reduced anxiety-like behavior and potentiated context-based fear responses, phenocopying the effects of lesions to septal projections to the mHb.
These results uncover an unexpected function for GFRα1 in the maintenance and function of adult glutamatergic synapses and reveal a potential new mechanism for regulating synaptic plasticity in the septo-habenulo-interpeduncular pathway and attuning of anxiety and fear behaviors.
The paper has been published in PLOS Biology
July 11th, 2021 Life- style change and anti-inflammatory interventions have only transient effects in obesity. It is not clear how benefits obtained by these treatments can be maintained longer term, especially during sustained high caloric intake. Constitutive ablation of the activin receptor ALK7 in adipose tissue enhances catecholamine signaling and lipolysis in adipocytes, and protects mice from diet-induced obesity.
In this study, we investigated the consequences of conditional ALK7 ablation in adipocytes of adult mice with pre- existing obesity. Although ALK7 deletion had little effect on its own, it synergized strongly with a transient switch to low- fat diet (life-style change) or anti-inflammatory treatment (Na-salicylate), resulting in enhanced lipolysis, increased energy expenditure, and reduced adipose tissue mass and body weight gain, even under sustained high caloric intake. By themselves, diet- switch and salicylate had only a temporary effect on weight gain. Mechanistically, combination of ALK7 ablation with either treatment strongly enhanced the levels of β3-AR, the main adrenergic receptor for catecholamine stimulation of lipolysis, and C/EBPα, an upstream regulator of β3-AR expression. These results suggest that inhibition of ALK7 can be combined with simple interventions to produce longer- lasting benefits in obesity.
The paper has been published in The FASEB Journal.
December 1st, 2020 A prevalent model of Alzheimer’s disease (AD) pathogenesis postulates the generation of neurotoxic fragments derived from the amyloid precursor protein (APP) after its internalization to endocytic compartments. The molecular pathways that regulate APP internalization and intracellular trafficking in neurons are incompletely understood.
In this paper, we report that 5xFAD mice, an animal model of AD, expressing signaling-deficient variants of the p75 neurotrophin receptor (p75NTR) show greater neuroprotection from AD neuropathology than animals lacking this receptor. p75NTR knock-in mice lacking the death domain or transmembrane Cys259 showed lower levels of Aβ species, amyloid plaque burden, gliosis, mitochondrial stress and neurite dystrophy than global knock-outs. Strikingly, long-term synaptic plasticity and memory, which are completely disrupted in 5xFAD mice, were fully recovered in the knock-in mice. Mechanistically, we found that p75NTR interacts with APP at the plasma membrane and regulates its internalization and intracellular trafficking in hippocampal neurons. Inactive p75NTR variants internalized considerably slower than wild type p75NTR and showed increased association with the recycling pathway, thereby reducing APP internalization and colocalization with BACE1, the critical protease for generation of neurotoxic APP fragments, favoring non-amyloidogenic APP cleavage. These results reveal a novel pathway that directly and specifically regulates APP internalization, amyloidogenic processing and disease progression, and suggest that inhibitors targeting the p75NTR transmembrane domain may be an effective therapeutic strategy in AD.
The paper has been published in The EMBO Journal.
August 3rd, 2020 Alvaro Carrier Ruiz received a PhD from the Faculty of Medicine of the University of Tokyo, specializing in Neurophysiology, under the supervision of Professor Masanobu Kano. Originally from Brazil, Alvaro joins our KI team to pursue studies on the physiological relevance of ALK4 signaling for brain function.
May 5th, 2020 Adaptation to nutrient availability is crucial for survival. Upon nutritional stress, such as during prolonged fasting or cold exposure, organisms need to balance the feeding of tissues and the maintenance of body temperature. Mechanisms regulating the adaptation of brown adipose tissue (BAT), a key organ for non-shivering thermogenesis, to variations in nutritional state have been unknown.
In this new paper, we report that specific deletion of the activin receptor ALK7 in BAT resulted in fasting-induced hypothermia due to exaggerated catabolic activity in brown adipocytes. After overnight fasting, BAT lacking ALK7 showed increased expression of genes responsive to nutrient stress, including the upstream regulator KLF15, aminoacid catabolizing enzymes, notably proline dehydrogenase (POX), and adipose triglyceride lipase (ATGL), as well as markedly reduced lipid droplet size. In agreement with this, ligand stimulation of ALK7 suppressed POX and KLF15 expression in both mouse and human brown adipocytes. Treatment of mutant mice with the glucocorticoid receptor antagonist RU486 restored KLF15 and POX expression levels in mutant BAT, suggesting that loss of BAT ALK7 results in excessive activation of glucocorticoid signaling upon fasting. These results reveal a novel signaling pathway downstream of ALK7 which regulates the adaptation of BAT to nutrient availability by limiting nutrient stress-induced overactivation of catabolic responses in brown adipocytes
The paper has been published in eLife.
February 12th, 2020 Nyantakyi Samuel Agyei obtained a PhD at the Department of Pharmacy of the National University of Singapore, which focused on identifying novel small molecules selectively targeting the cell membrane of Mycobacterium tuberculosis. Samuel’s core expertise is in multi-step syntheses of small molecules, analyses of structure-activity relationships to improve compound quality, and building and managing compound libraries.
Samuel joins our KI team to develop medicinal chemistry approaches to modify and improve on candidate small molecules capable of modulating the activities of various receptors, including our recently discovered compounds targeting p75NTR, based on our article in Cell Chemical Biology.
January 9th, 2020 In this new paper, we report that CD137, a cell surface protein used in several studies as a marker for beige adipocytes, is undetectable at the protein level in beige adipocytes in vivo or in vitro, and its expression is not upregulated by adrenergic stimulation or cold exposure, as expected for a beige cell marker. Moreover, CD137 knock-out mice showed elevated levels of thermogenic markers, including UCP1, increased numbers of beige adipocyte precursors, and expanded UCP1-expressing cell clusters in inguinal WAT after chronic cold exposure. CD137 knock-out mice also showed enhanced cold resistance. These results indicate that CD137 functions as a negative regulator of “browning” in white adipose tissue, and call into question the use of this protein as a functional marker for beige adipocytes.
The paper has just been published in The Journal of Biological Chemistry.
January 1st, 2020 Starting in January 2020, new twin laboratories dedicated to studies of growth factor receptor signaling and physiology will be established at the McGovern Institute of the School of Life Sciences in Peking University, and the Chinese Institute for Brain Research in Beijing, China. The research activities of the PKU and CIBR labs will run in parallel to and complement with those ongoing at the KI and NUS laboratories. The initial focus of the new labs will be on studies of death receptor signaling in neurodegeneation, metabolic regulation by activin receptors ALK4 and ALK7, and control of brain microvasculature integrity and function by neurotrophin signaling. PhD students, postdoctoral fellows and lab technicians are being �recruited for the new Beijing laboratories. Follow developments in the PKU and CIBR labs at HERE.
November 1st, 2019 In this new paper, we report how the activin receptor ALK4 coordinates signaling by activin ligands with intrinsic transcriptional programs driven by SATB1 to regulate the development of somatostatin interneurons in the developing mouse neocortex.
Although the role of transcription factors in fate specification of cortical interneurons is well established, how these interact with extracellular signals to regulate interneuron development is poorly understood. Here we show that the activin receptor ALK4 is a key regulator of the specification of somatostatin interneurons. Mice lacking ALK4 in GABAergic neurons of the medial ganglionic eminence (MGE) showed marked deficits in distinct subpopulations of somatostatin interneurons from early postnatal stages of cortical development. Specific losses were observed among distinct subtypes of somatostatin+/Reelin+ double-positive cells, including Hpse+ layer IV cells targeting parvalbumin+interneurons, leading to quantitative alterations in the inhibitory circuitry of this layer. Activin-mediated ALK4 signaling in MGE cells induced interaction of Smad2 with SATB1, a transcription factor critical for somatostatin interneuron development, and promoted SATB1 nuclear translocation and repositioning within the somatostatin gene promoter. These results indicate that intrinsic transcriptional programs interact with extracellular signals present in the environment of MGE cells to regulate cortical interneuron specification.
The paper has just been published in The Journal of Cell Biology .
Read the full paper HERE.
August 29th, 2019 UPDATE : the position has been filled.
We are seeking a talented and enthusiastic researcher with expertise in medicinal chemistry and chemical biology to advance drug discovery efforts and investigations on death receptor signaling and physiology in cancer and neuronal cells. The research entails medicinal chemistry studies on small molecules targeting the p75NTR death receptor identified in our ongoing screens, with the objective to improve the pharmacokinetic and biological properties of chemical leads. Please see Goh et al. (2018) Cell Chemical Biology 25, 1485–1494 for a representative publication of this work.
Candidates will be accepted at the postdoctoral level with a PhD awarded within the last 3 years. Strong and documented expertise in medicinal chemistry and chemical biology is a requirement for consideration. The successful candidate is expected to be sufficiently independent to formulate questions, design experiments and perform research. Excellent command of the English language is reuqired.
Applications, including CV (no diploma copies needed at this time please), list of publications and statement of research interests should be sent by email to Prof. Carlos Ibanez ( ) and through the job web page of Karolinska Institute. Applicants should arrange to have at least two confidential letters of reference sent independently by referees to this email address. Funding is available for an initial period of 3 to 4 years. ) and through the job web page of Karolinska Institute. Applicants should arrange to have at least two confidential letters of reference sent independently by referees to this email address. Funding is available for an initial period of 3 to 4 years.
Deadline for application is 29 September 2019.
August 8th, 2019 UPDATE September 2019: The position has been filled.
We are seeking talented and enthusiastic researchers with expertise in molecular and cellular neuroscience to advance investigations on the functions and mechanisms of growth factor receptor signaling and physiology in the nervous system. The research entails studies of different receptor systems in neuronal and nervous system function using molecular methods, cell culture models and mutant mice carrying specific mutations in these receptors.
Candidates will be accepted at the postdoctoral level with a PhD awarded preferably within the last 3 years. Strong and documented expertise in molecular, cellular and histological methods of analysis as applied to studies of mouse models in the area of neuroscience is an absolute requirement for consideration. Successful applicants shall be well versed in mouse brain neuroanatomy and physiology. Expertise in analysis of mouse behavior will be an additional advantage. The successful candidate is expected to be sufficiently independent to formulate questions, design experiments and perform research. For more information and publications, please look HERE in our lab website.
Applications, including CV, list of publications and statement of research interests should be sent through the job web page of Karolinska Institute: Applicants should arrange to have at least two confidential letters of reference sent independently by referees to that email address.
Deadline for application is 15 September 2019.
June 10th, 2019 Michael Saleeb obtained a PhD in Organic Chemistry at Umeå University in Sweden in September 2018, under the direction of Prof. Mikael Elofsson. His PhD thesis was focused on the identification of novel antibacterial compounds against Chlamydia trachomatis & Pseudomonas aeruginosa infections. During his PhD, Michael developed expertise in chemical synthesis including C-C and C-X metal-catalyzed cross coupling, C-H insertion reactions, various heterocyclic and polyphenol syntheses. Michael joins our KI team to develop medicinal chemistry approaches to modify and improve on candidate small molecules capable of modulating the activities of various receptors, including our recently discovered compounds targeting p75NTR, based on our article in Cell Chemical Biology.
April 1st, 2019 Qiang Zhang obtained his PhD at the Karolinska Institute in 2018, under the direction of Prof. Klas Wiman. His PhD thesis was entitled “Understanding p53 structure and targeting mutant p53 for improved cancer therapy”. Qiang joins our KI team to work on the identification of small molecules capable of modulating the activities of the p75 neurotrophic receptor p75NTR, based on the platform described in our recent article in Cell Chemical Biology.
April 1st, 2019
Carlos Ibanez’s team returns to the Department of Neuroscience after 21 months under the administration of CMB. We are happy to be back, and hope to relocate to new space within Biomedicum later in the year.
February 1st, 2019 In this new paper, we report that abnormal TDP‐43 function culminate in impaired secretion of the neurotrophin BDNF, whose restoration is sufficient to rescue major disease phenotypes caused by aberrant TDP‐43 activity.
Aberrant function of the RNA‐binding protein TDP‐43 has been causally linked to multiple neurodegenerative diseases. Due to its large number of targets, the mechanisms through which TDP‐43 malfunction cause disease are unclear. Here, we report that knockdown, aggregation, or disease‐associated mutation of TDP‐43 all impair intracellular sorting and activity‐dependent secretion of the neurotrophin brain‐derived neurotrophic factor (BDNF) through altered splicing of the trafficking receptor Sortilin. Adult mice lacking TDP‐43 specifically in hippocampal CA1 show memory impairment and synaptic plasticity defects that can be rescued by restoring Sortilin splicing or extracellular BDNF. Human neurons derived from patient iPSCs carrying mutated TDP‐43 also show altered Sortilin splicing and reduced levels of activity‐dependent BDNF secretion, which can be restored by correcting the mutation. We propose that major disease phenotypes caused by aberrant TDP‐43 activity may be explained by the abnormal function of a handful of critical proteins, such as BDNF.
The paper has just been published in The EMBO Journal.
Read the full paper HERE.
January 11th, 2019 Ana Osorio Oliveira obtained her PhD at the Gladstone Institute, University of California San Francisco , USA, in 2015, under the direction of Profs. Paul Muchowski and Steven Finkbeiner. She performed postdoctoral studies at Stanford University under the direction of Dr. Xinnan Wang. Ana joins our KI team to work on studies of neurotrophic factor signaling and function in the adult nervous system.
November 16th, 2018 The Saturday edition of the The Straits Times publishes today an interview with Carlos Ibanez highlighting the identification a small molecule targeting the transmembrane domain of death receptor p75NTR that induces melanoma cell death and reduces tumor growth (reported in our paper published in Cell Chemical Biology). Read the full article here. Also newspaper Lianhe Zaobao (Chinese Daily) carried a story (in Chinese) on the discovery.
November 13th, 2018
Cancerfonden, the largest research funding charity organization in Sweden, today decided to award a special grant to Carlos Ibanez lab for drug discovery efforts targeting the p75 neurotrophin receptor in melanoma and glioma, based on our pilot study recently published in Cell Chemical Biology.
October 4th, 2018 In this new paper, we have used a novel chemical biology approach to identify a small molecule targeting the transmembrane domain of death receptor p75NTR that induces melanoma cell death and reduces tumor growth
Small molecules offer powerful ways to alter protein function. However, most proteins in the human proteome lack small-molecule probes, including the large class of non-catalytic transmembrane receptors, such as death receptors. We hypothesized that small molecules targeting the interfaces between transmembrane domains (TMDs) in receptor complexes may induce conformational changes that alter receptor function. Applying this concept in a screening assay, we identified a compound targeting the TMD of death receptor p75NTR that induced profound conformational changes and receptor activity. The compound triggered apoptotic cell death dependent on p75NTR and JNK activity in neurons and melanoma cells, and inhibited tumor growth in a melanoma mouse model. Due to their small size and crucial role in receptor activation, TMDs represent attractive targets for small-molecule manipulation of receptor function.
The paper has just been published in Cell Chemical Biology.
Read the full paper HERE.
July 24th, 2018 In this new paper, we show how intracellular effectors RIP2 and TRAF6 compete for binding to the p75NTR intracellular domain to regulate cell death of cerebellar granule neurons.
Cerebellar granule neurons (CGNs) undergo programmed cell death during the first postnatal week of mouse development, coincident with sustained expression of the death receptor p75NTR. Although ablation of p75NTR did not affect CGN cell death, deletion of the downstream effector RIP2 significantly increased CGN apoptosis, resulting in reduced adult CGN number and impaired behaviors associated with cerebellar function. Remarkably, CGN death was restored to basal levels when p75NTR is deleted in RIP2-deficient mice. We found that RIP2 gates the signaling output of p75NTR by competing with TRAF6 for binding to the receptor intracellular domain. In CGNs lacking RIP2, more TRAF6 was associated with p75NTR, leading to increased JNK-dependent apoptosis. In agreement with this, pharmacological inhibition or genetic ablation of TRAF6 restored cell death levels in CGNs lacking RIP2. These results revealed an unexpected mechanism controlling CGN number and highlight how competitive interactions govern the logic of death receptor function.
The paper has just been published in Cell Reports.
Read the full paper HERE.
May 30th, 2018 In this new paper, we show how the GDNF receptor GFRα1 functions cell-autonomously in subpopulations of olfactory bulb interneuron precursors to regulate their generation and allocation in the mammalian olfactory bulb.
GFRα1, a receptor for glial cell line-derived neurotrophic factor (GDNF), is critical for the development of the main olfactory system. The olfactory bulb (OB) of Gfra1 knockout mice showed significant reductions in the number of olfactory sensory neurons, mitral and tufted cells, as well as all major classes of OB GABAergic interneurons. However, the latter did not express significant levels of GFRα1, leaving the mechanism of action of GFRα1 in OB interneuron development unexplained. We have found that GFRα1 is highly expressed in the precursor cells that give rise to all major classes of OB interneurons, but is downregulated as these neurons mature. Conditional ablation of GFRα1 in embryonic GABAergic cells recapitulated the cell losses observed in global Gfra1 knockouts at birth. GFRα1 was also required for the sustained generation and allocation of OB interneurons in adulthood. Conditional loss of GFRα1 altered the migratory behaviour of neuroblasts along the rostral migratory stream (RMS) as well as RMS glial tunnel formation. Together, these data indicate that GFRα1 functions cell-autonomously in subpopulations of OB interneuron precursors to regulate their generation and allocation in the mammalian OB.
The paper has just been published in Biology Open.
Read the full paper HERE.
April 27th, 2018 Today is the big day. After many years of planning and construction, all the preclinical departments of Karolinska Institute move into a huge, brand-new building called BIOMEDICUM. Very handsome on the inside, less so on the outside. We are just hoping for a smooth transition.
December 10th, 2017 Presentation Speech by Professor Carlos Ibáñez, Member of the Nobel Assembly at the Karolinska Institute, Member of the Nobel Committee for Physiology or Medicine, at the award ceremony of the Nobel Prize, 10 December 2017.
August 1st, 2017 Shounak Baksi obtained his PhD at the University of Calcutta, India, in September 2009. His thesis work focused on studies of alterations in Growth Factor Receptor Protein Binding Protein 2 (Grb2) signaling in Huntington’s disease cell model. His postdoctoral studies in Case Western Reserve University, Cleveland, OH, under the direction of Dr. Neena Singh, focused on the role of Parkinson’s disease protein alpha synuclein in retinal and brain iron homeostasis. Shounak established the role of alpha synuclein in the process of transferrin receptor endocytosis. Shounak joins the KI team to pursue studies of p75 signaling mutant mice.
August 1st, 2017 Carlos Ibanez’s team is now under the Department of Cell and Molecular Biology (CMB) of Karolinska Institute. Although the group was since 1996 under the Department of Neuroscience, its laboratories were already located at CMB space since 2004. This administrative change completes the integration of the Ibanez team in the CMB organization, in preparation for the big move to the new Biomedicum building next year.
June 14th, 2017 In this new paper, we show how the GDNF regulates survival of molecular layer interneurons in the cerebellum to control normal cerebellar motor learning. The paper has just been published in Cell Reports.
The role of neurotrophic factors as endogenous survival proteins for brain neurons remains contentious. In the cerebellum, the signals controlling survival of molecular layer interneurons (MLIs) are unknown, and direct evidence for the requirement of a full complement of MLIs for normal cerebellar function and motor learning has been lacking. Here, we show that Purkinje cells (PCs), the target of MLIs, express the neurotrophic factor GDNF during MLI development and survival of MLIs depends on GDNF receptors GFRα1 and RET. Conditional mutant mice lacking either receptor lose a quarter of their MLIs, resulting in compromised synaptic inhibition of PCs, increased PC firing frequency, and abnormal acquisition of eyeblink conditioning and vestibulo-ocular reflex performance, but not overall motor activity or coordination. These results identify an endogenous survival mechanism for MLIs and reveal the unexpected vulnerability and selective requirement of MLIs in the control of cerebellar-dependent motor learning.
Read the full paper HERE.
|
|